
Iron atoms are oxidized to become iron(II) ions The more reactive metal is oxidized The less reactive metal is reduced For example, Chemical Equation: Fe (s) + CuSO4 (aq) FeSO4 (aq) + Cu (s) Ionic Equation: Fe (s) + Cu2+(aq) Fe2+ (aq) + Cu (s) REDOX reaction! Copper(II) ions are reduced to become copper atoms Displacement Reactions are Redox Reactions

Iron Nail in Copper(II) Sulphate Solution We say that… Iron has displaced copper from the copper(II) sulphate solution. Iron(II) sulphate solution is pale green in colour.

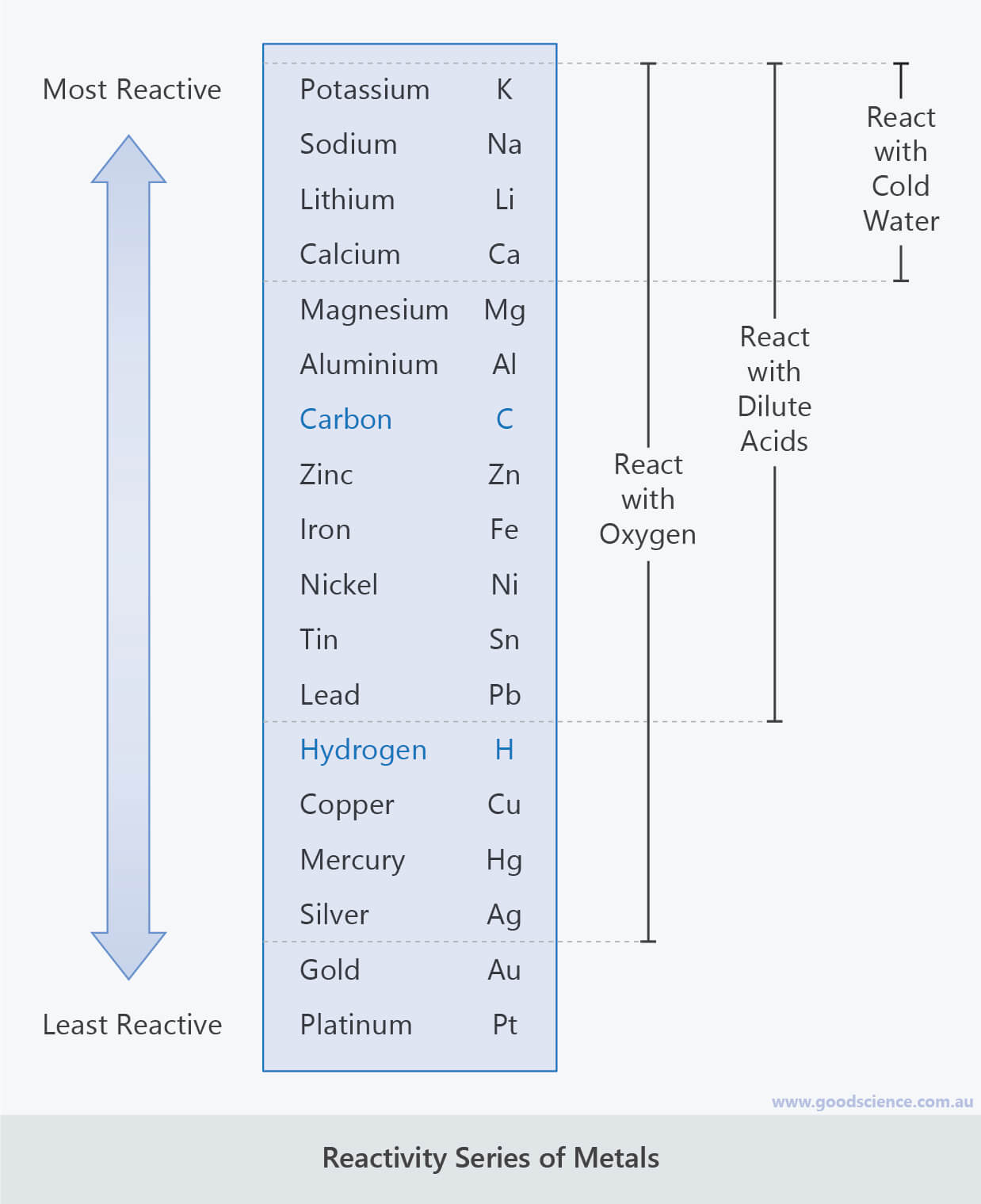
#Metal reactivity series series
2K(s) + 2HCl(l) 2KCl(aq) + H2(g) The more vigorous the reaction The higher it is in the reactivity series The more reactive is the metal Recap: Chemical Properties of Metals Reaction with Hydrochloric Acid Metal + Hydrochloric Acid Salt + Hydrogen Gas Eg. Mg(s) + H2O(g) MgO(s) + H2(g) Which metals only react with steam? Recap: Chemical Properties of Metals Reaction with Water (Steam) Metal + Steam Metal Oxide + Hydrogen Gas Eg. 2Na(s) + 2H2O(l) 2NaOH(aq) + H2(g) Which are some metals that react with cold water? Recap: Chemical Properties of Metals Reaction with Water (Cold Water) Metal + Cold Water Metal Hydroxide + Hydrogen Gas Eg. Thermal stability & reactivity Reactivity Series of Metals.Reaction of Metals: Displacement Reactions.Metal reaction with cold water & steam.The less reactive metal is a better electron acceptor than the more reactive metal, thus the less reactive metal is reduced.Reactivity Series of Metals Reactions of Metals Effect of Heat on Metal Carbonates.This is because more reactive metals lose electrons and form ions more readily than less reactive metals, making them better reducing agents.Any metal will displace another metal that is below it in the reactivity series from a solution of one of its salts.The reducing agent in the reaction is magnesium:Ĭopper oxide + magnesium → copper + magnesium oxideĭisplacement reactions between metals and aqueous solutions of metal salts.The less reactive metal, in this case copper, ends up in elemental form as it is reduced.As magnesium is above copper in the reactivity series, magnesium is more reactive so can displace copper from its oxide.It is possible to reduce copper(II) oxide by heating it with magnesium.This means that a more reactive metal can displace a less reactive metal from its oxide by heating.The reactivity of metals increases going up the reactivity series.
